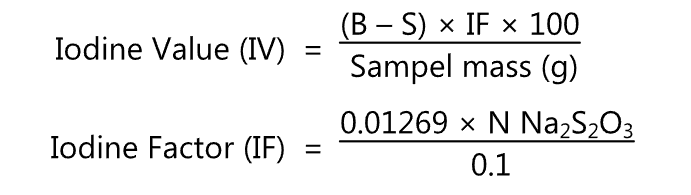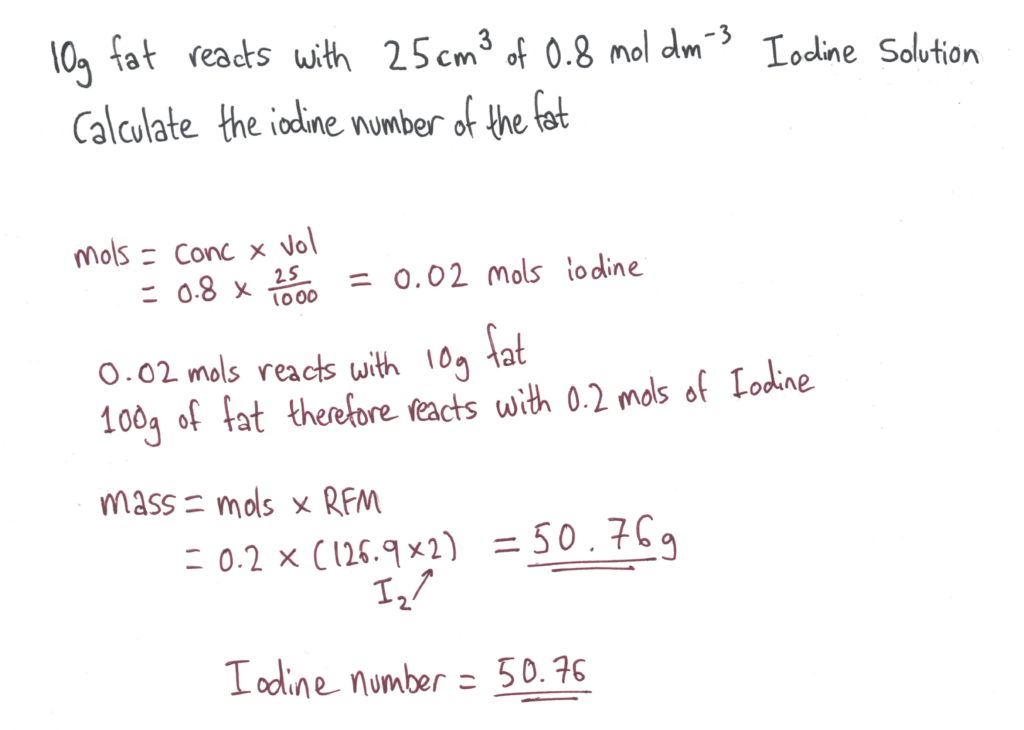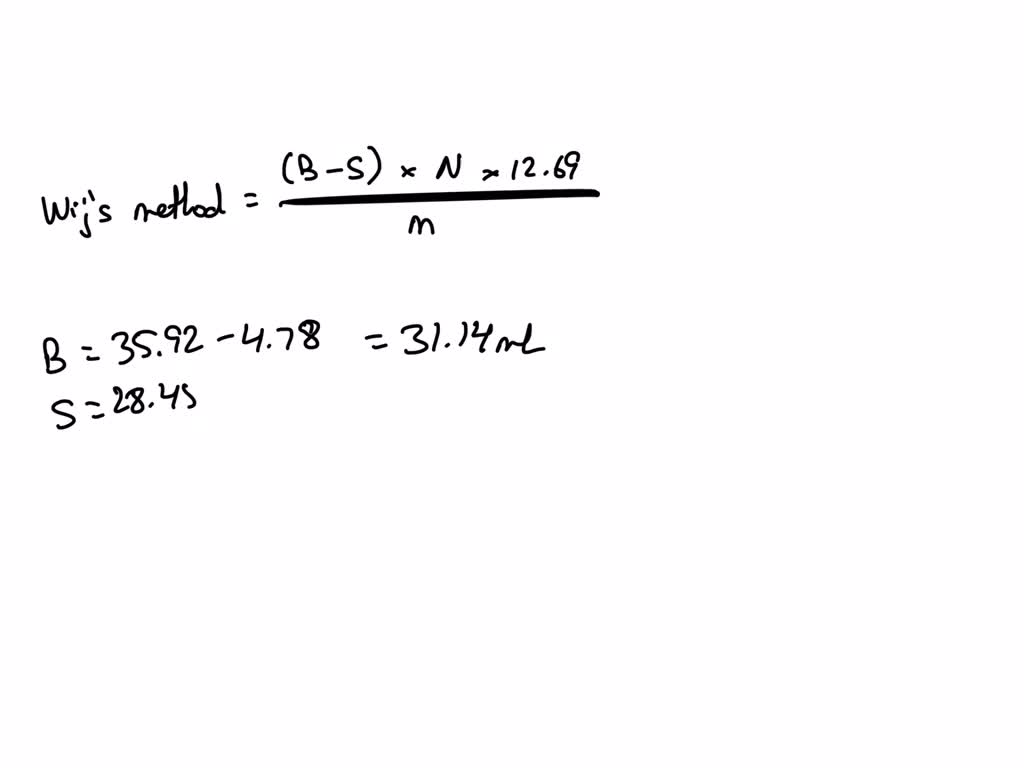
SOLVED: Analysis for the determination of the iodine number (IN) of an unknown sample was conducted using Wij's method. Calculate the iodine number using the following data: Sample weight: 10 grams Titration

Iodine number or iodine value - The iodine number or iodine value of a fat or oil is defjned as the - Studocu
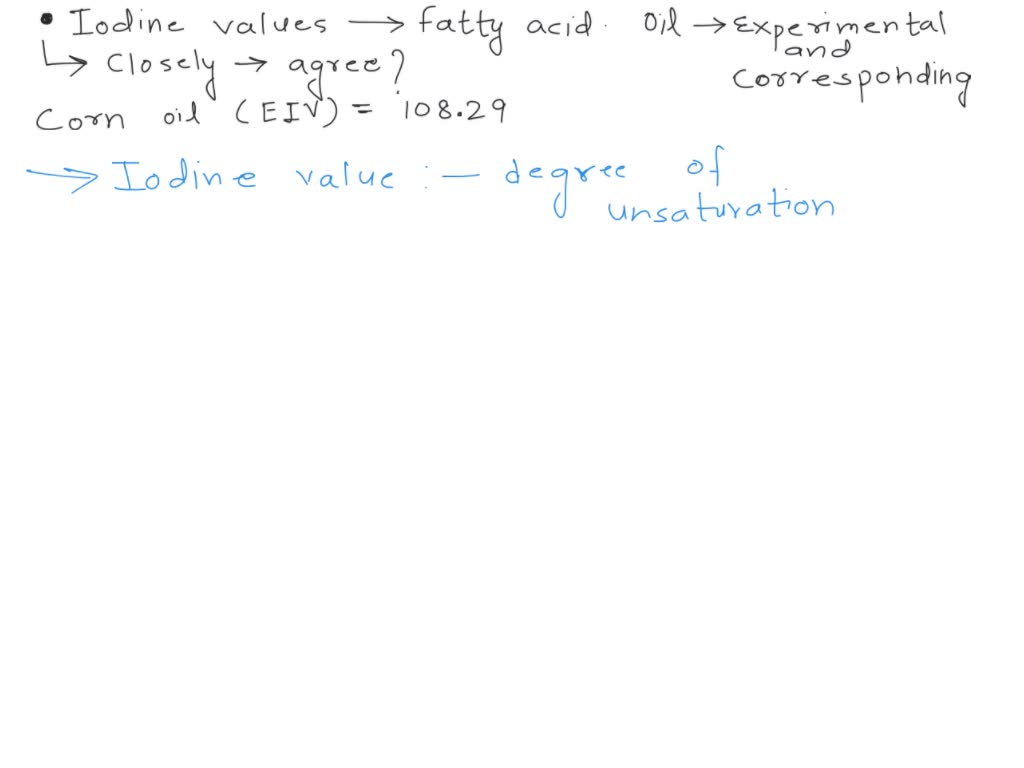
SOLVED: Iodine values can be calculated from the fatty acid composition of an oil or fat. Find, in the literature, the fatty acid composition of the oil for which you have determined

SOLVED: Hanus Method for Determination of Iodine Number Weigh 0.3-1.0 grams of an oil into a flask. Add 10 mL of anhydrous chloroform. Add 30.0 mL of Hanus reagent to the flask.

Iodine number, Saponification number, Acid Number, Reichert-Meissl (RM) number & Rancidity of Oil/Fats | The Pharmapedia

A green potentiometric method for the determination of the iodine number of biodiesel - ScienceDirect

Iodine value: Definition, Principle, Procedure, Formula, and 2 Reliable Applications - Chemistry Notes
![PDF] Simple spectrophotometric method for determination of iodine value of vegetable oils | Semantic Scholar PDF] Simple spectrophotometric method for determination of iodine value of vegetable oils | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3932fa6fdb43b2a747ea63a758bc47f7409684e0/6-Table3-1.png)