Understanding the Stabilizing Effect of Histidine on mAb Aggregation: A Molecular Dynamics Study | Molecular Pharmaceutics
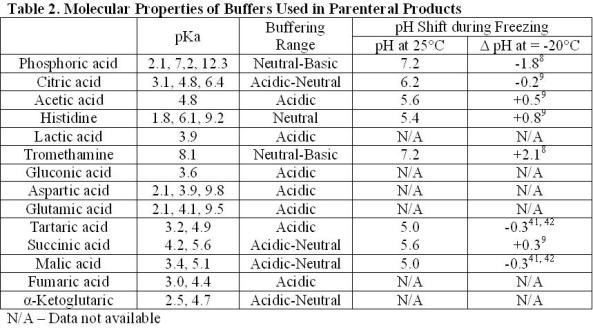
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review
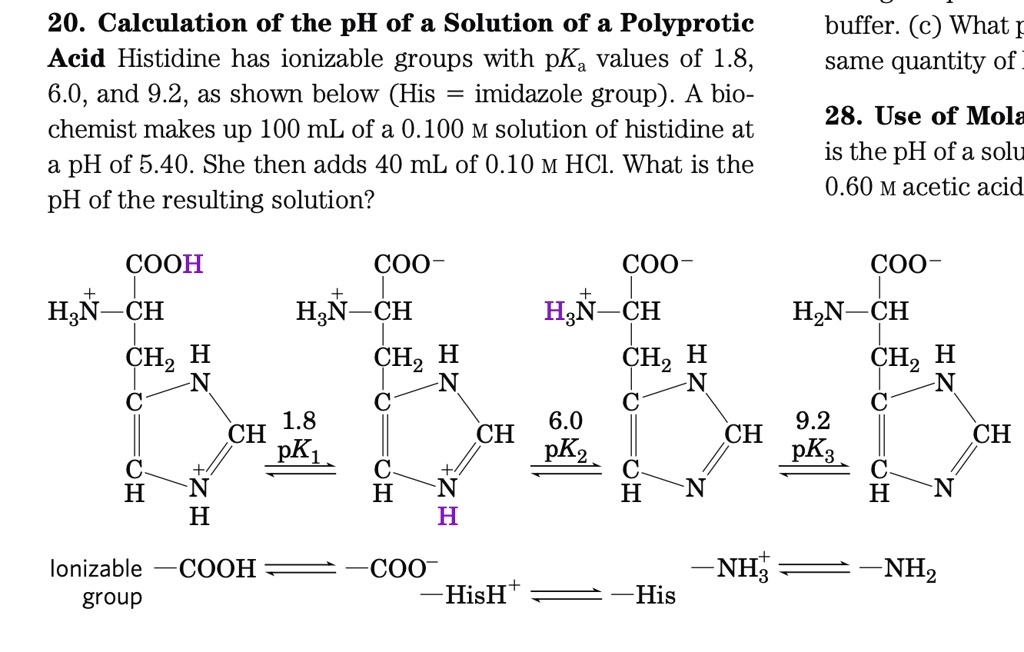
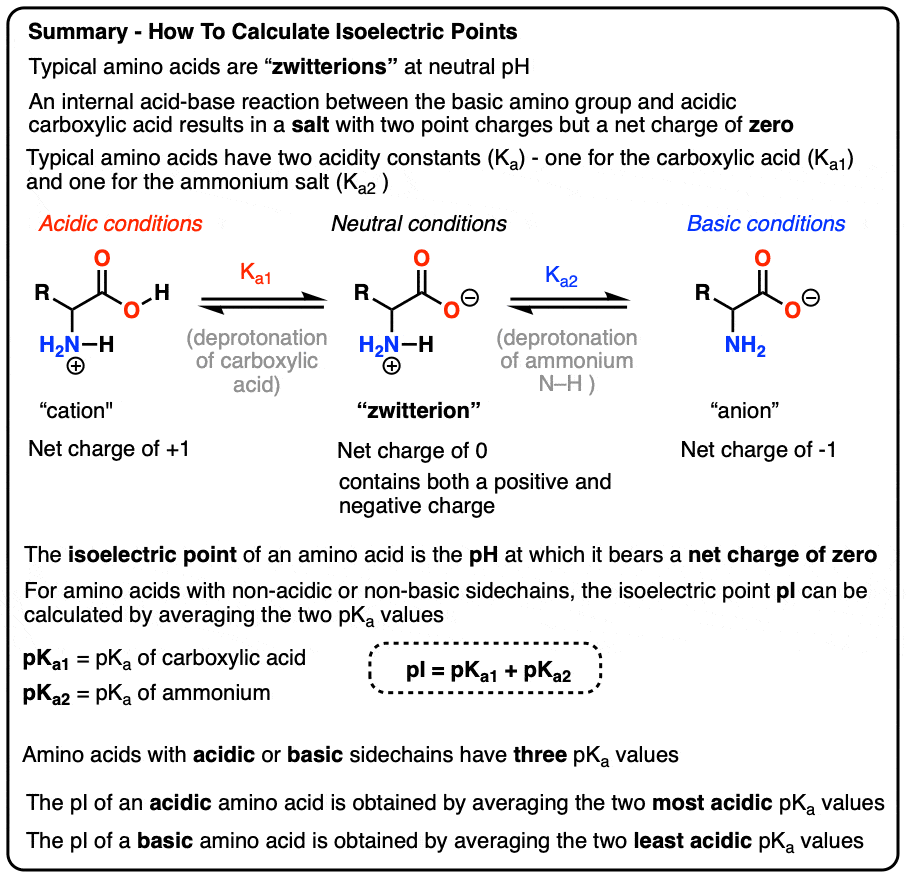
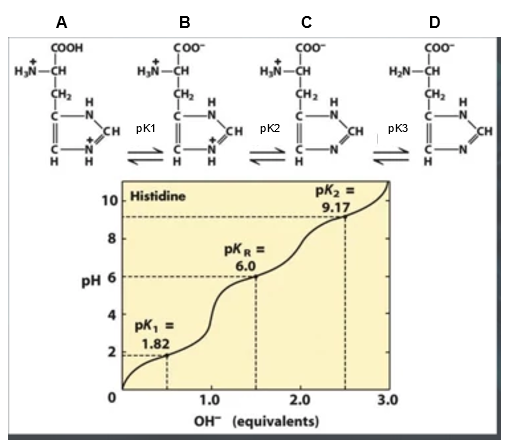
SOLVED: 20. Calculation of the pH of a Solution of a Polyprotic Acid Histidine has ionizable groups with pKa values of 1.8, 6.0, and 9.2, as shown below (His imidazole group). A

Change in pH as a function of temperature for histidine acetate buffer... | Download Scientific Diagram

SOLVED: 20. Calculation of the pH of a Solution of a Polyprotic Acid Histidine has ionizable groups with pKa values of 1.8, 6.0, and 9.2, as shown below (His imidazole group). A
Call: H2020-MSCA-ITN-2015 Topic: MSCA-ITN-2015-ETN Project / GA number: 675074 Project full title: Protein-excipient Interactio