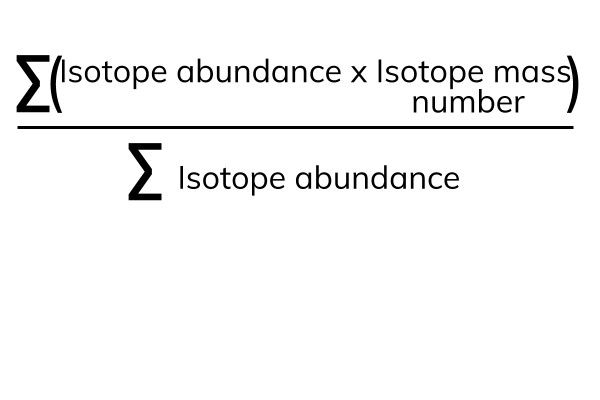
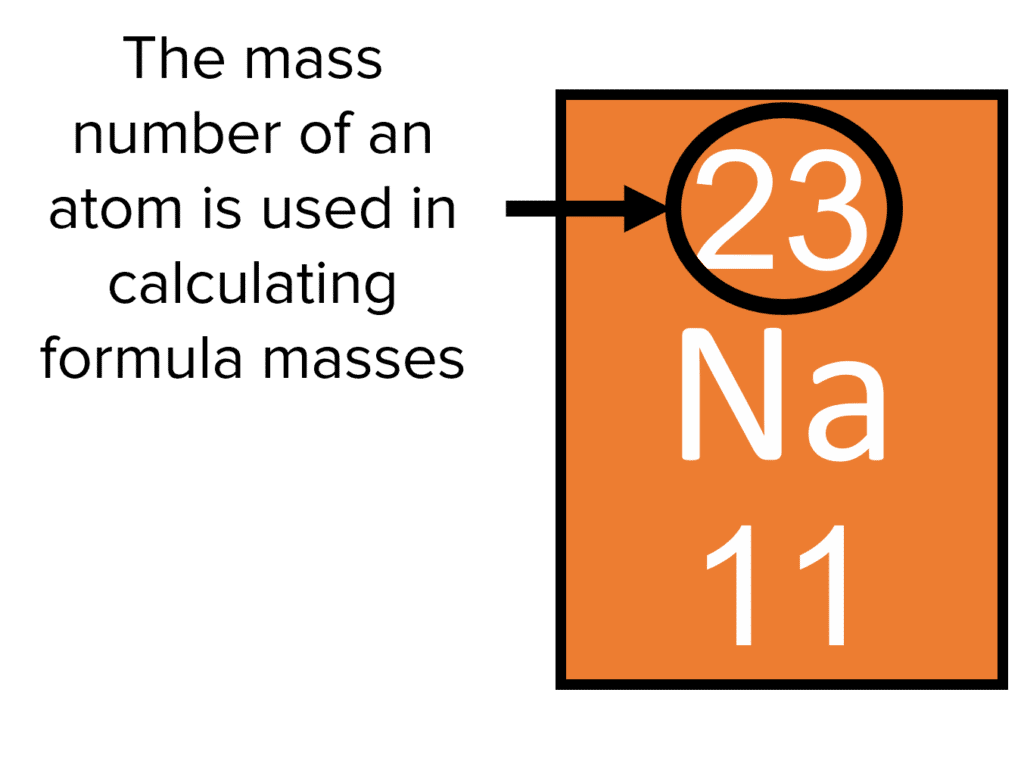![Question 4 (a) Calculate Relative atomic mass the following: (i) SO2 [S=32 0=16] (ii) HSO4 [H=1 S=32 0=16] (iii) Mg3N2 [Mg=24 N = 14] (iv) AlCl3 [Al=27 CI=35.5] (v) P2O5 [P=31 0=16] Question 4 (a) Calculate Relative atomic mass the following: (i) SO2 [S=32 0=16] (ii) HSO4 [H=1 S=32 0=16] (iii) Mg3N2 [Mg=24 N = 14] (iv) AlCl3 [Al=27 CI=35.5] (v) P2O5 [P=31 0=16]](https://toppr-doubts-media.s3.amazonaws.com/images/9198340/812985f2-af75-4222-a9fb-8b231d50fcf4.jpg)
Question 4 (a) Calculate Relative atomic mass the following: (i) SO2 [S=32 0=16] (ii) HSO4 [H=1 S=32 0=16] (iii) Mg3N2 [Mg=24 N = 14] (iv) AlCl3 [Al=27 CI=35.5] (v) P2O5 [P=31 0=16]
![A-level chem/math: relative atomic number] Why should we use the percentage of mass as abundance? : r/HomeworkHelp A-level chem/math: relative atomic number] Why should we use the percentage of mass as abundance? : r/HomeworkHelp](https://preview.redd.it/esr10skr4xc91.png?width=1397&format=png&auto=webp&s=b6da051206a92a6636081e34ba2520936133dd3a)
A-level chem/math: relative atomic number] Why should we use the percentage of mass as abundance? : r/HomeworkHelp

Questions for calculating relative atomic mass | Teaching Resources | Relative atomic mass, Chemistry worksheets, Chemistry notes

![Relative atomic mass_&_mass_spectrometry[1][1] | PPT Relative atomic mass_&_mass_spectrometry[1][1] | PPT](https://image.slidesharecdn.com/relativeatomicmassmassspectrometry11-111114171236-phpapp01/85/relative-atomic-massmassspectrometry11-33-320.jpg?cb=1668100786)