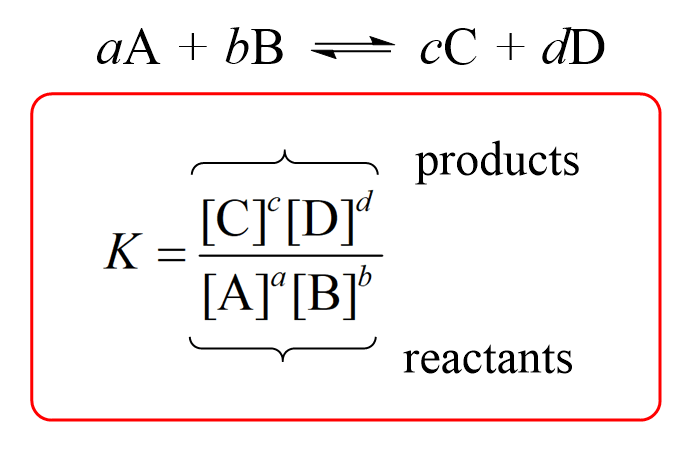
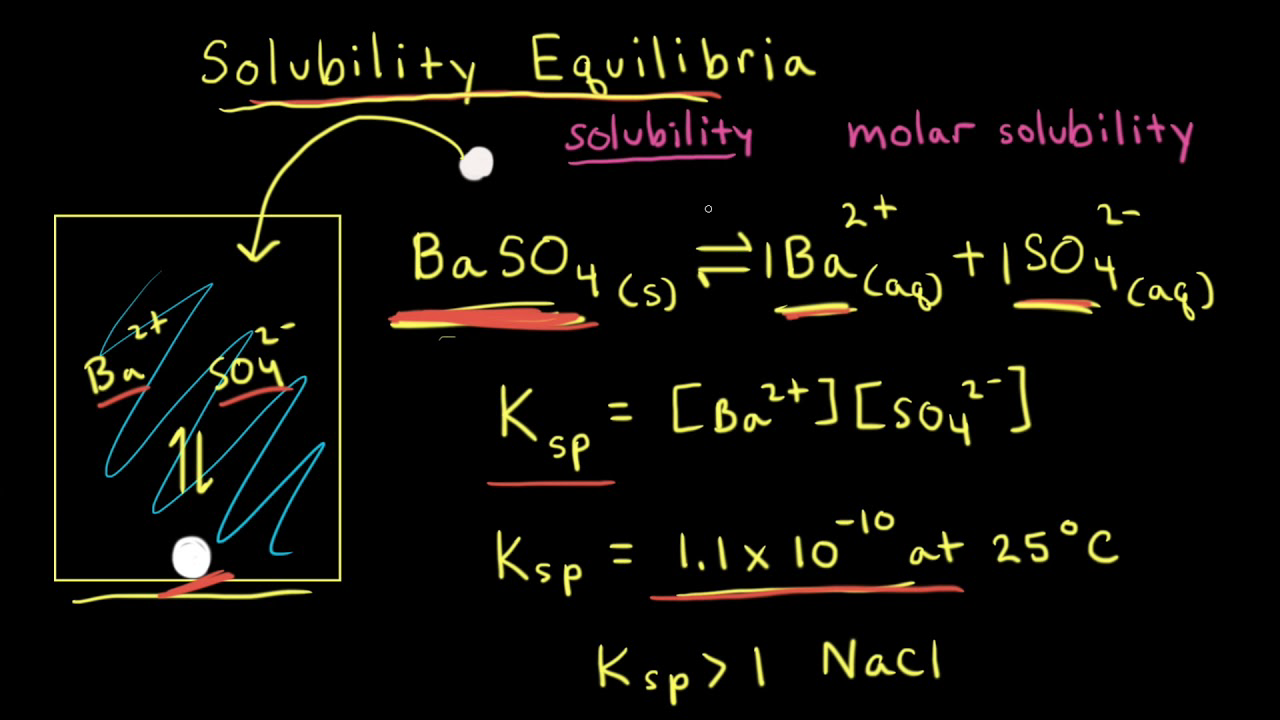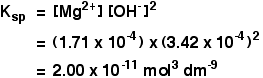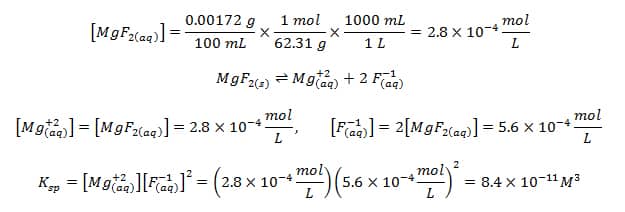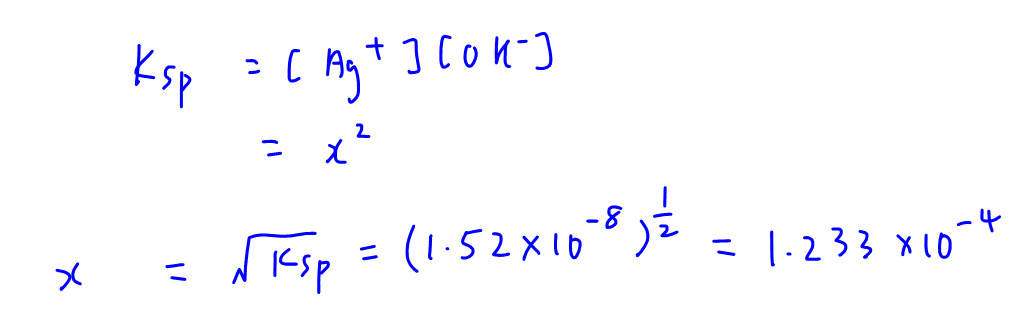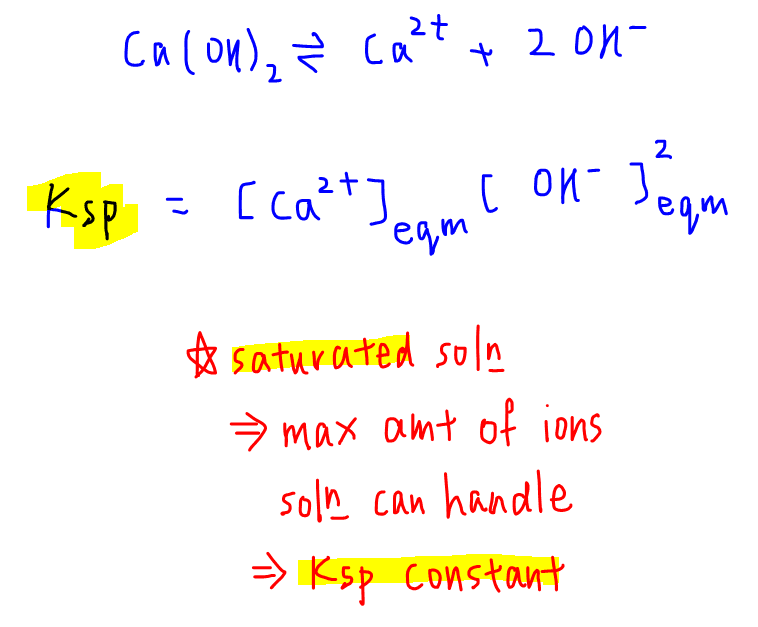Calculate the solubility product of AgCl if the solubility of the salt in saturated solution at 298 K is 0.00198 g L^(-1).

Calculate the solubility product constant of AgI from the following values of standard electrode potentials. E(Ag^(+)//Ag)^(@)=0.80 volt and E(I//AgI//Ag)^(@)=-0.15 volt at 25^(@)C
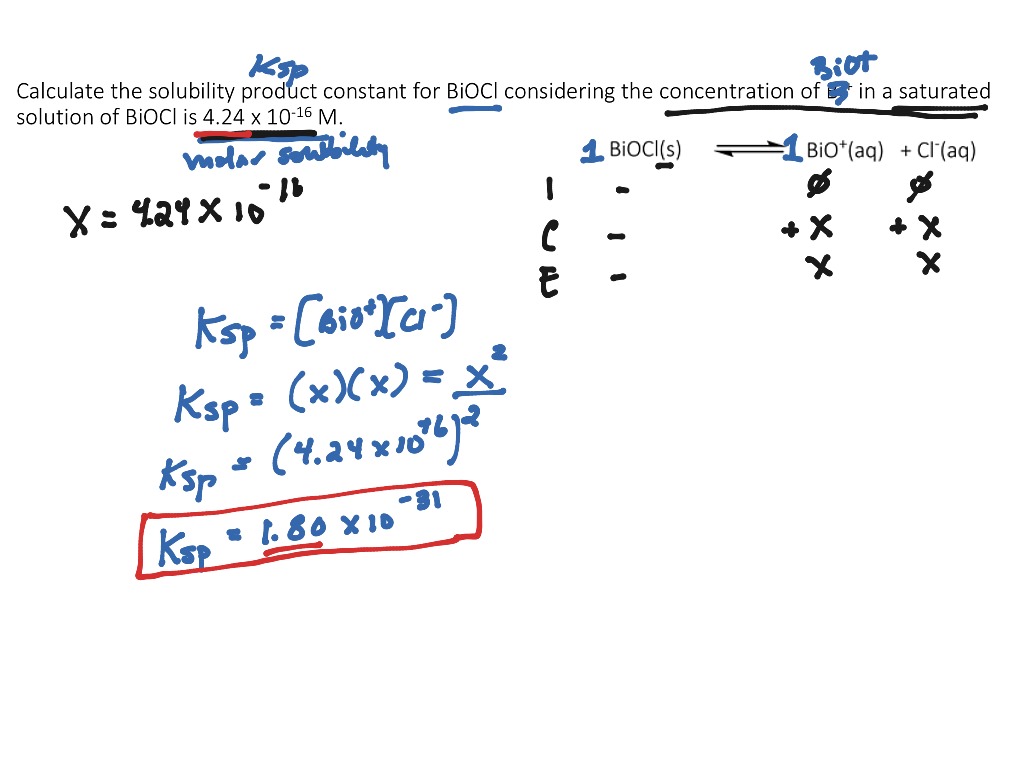
Calculating the Solubility Product Constant (Ksp) for an Insoluble Salt | Science, Chemistry | ShowMe
![The solubility product of PbCl2 at 298 K is 1.7 × 10^-5 . Calculate the solubility of PbCl2 in g/lit at 298K .Atomic weights : [ Pb = 207 and Cl = 35.5 ] The solubility product of PbCl2 at 298 K is 1.7 × 10^-5 . Calculate the solubility of PbCl2 in g/lit at 298K .Atomic weights : [ Pb = 207 and Cl = 35.5 ]](https://d1hhj0t1vdqi7c.cloudfront.net/v1/SFkyRXpPWTdqMnc=/sd/)